Abstract
Background: Follicular lymphoma is an indolent disease with a continuous relapsing pattern and typically requires multiple lines of therapy. Novel therapies such as tisagenlecleucel are being investigated to improve outcomes. Primary analysis of the single-arm, multicenter, Phase II ELARA trial in r/r FL demonstrated that tisagenlecleucel resulted in high overall (ORR) and complete response rates (CRR), and prolonged progression-free survival (PFS) at a median follow-up of 11 months (mo). Here, we report updated efficacy results from the overall population at a median follow-up of 17 mo, and a subgroup analysis of pts with high-risk disease from the ELARA trial (NCT03568461).
Methods: Eligible adult pts had histologically confirmed r/r FL (grades 1-3A) after ≥2 lines of therapy or had relapsed after autologous stem cell transplant. Bridging therapy was allowed and was followed by disease evaluation before tisagenlecleucel infusion. Pts received tisagenlecleucel (0.6-6×10 8 CAR+ viable T cells) after lymphodepleting chemotherapy (fludarabine [25 mg/m 2] + cyclophosphamide [250 mg/m 2] QD for 3 d or bendamustine [90 mg/m 2] QD for 2 d). Endpoints included ORR, CRR, PFS, and duration of response (DOR). Descriptive efficacy subanalyses were performed for 9 high-risk subgroups, including prior hematopoietic stem cell transplant (HSCT), ≥5 prior lines of therapy, progression of disease within 24 mo from first immunochemotherapy (POD24), double-refractory disease, high Follicular Lymphoma International Prognostic Index (FLIPI) at study entry, high lactate dehydrogenase at baseline, high C-reactive protein (CRP) prior to infusion, radiological bulky disease (by GELF criteria), and high total metabolic tumor volume (TMTV; >510 cm 3) at baseline (median 155.32 cm 3; range 0.1-2470.4 cm 3). Descriptive subgroup analysis was supported by multivariate analysis to identify factors predictive of worse outcomes.
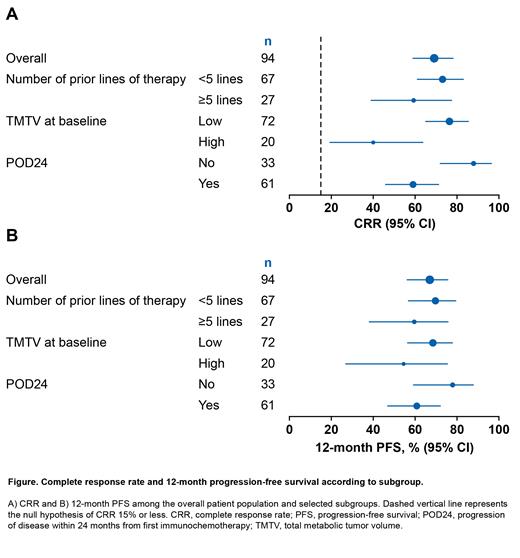
Results: As of March 29, 2021, 97 pts received tisagenlecleucel and 94 were evaluable for primary efficacy analysis (median follow-up 17 mo). High and durable responses were seen in the overall ELARA population (ORR 86.2%, CRR 69.1%, 9-mo DOR 76.0%, and 12-mo PFS 67.0%). In CR pts at 9 mo, PFS was 85.5% and estimated probability of remaining in response was 86.5%. Safety reflected known tisagenlecleucel profile; 48% of pts had CRS (majority were grade 1/2) and 11.3% had neurological events (3% grade ≥3). In the subgroup analysis, pts were stratified into risk groups. Efficacy (ORR, CRR) and durability of response were well maintained in all high-risk subgroups, except for POD24 (n=35), high TMTV (n=20), and ≥5 prior lines of therapy (n=27). Compared with corresponding low-risk subgroups, there was a numerical reduction in CRR for high-risk subgroups (POD24 59.0% vs 87.9%; high TMTV 40.0% vs 76.4%; ≥5 prior lines of therapy 59.3% vs 73.1%) (Figure). A reduction in 12-mo PFS was also identified for pts in these subgroups: POD24 (60.8% vs 77.9%), high baseline TMTV (54.5% vs 68.5%), and ≥5 prior lines of therapy (59.6% vs 69.7%). Evaluating the disease characteristics of the high TMTV subgroup compared with low TMTV, high TMTV was associated with a higher incidence of bulky disease (58.3% vs 90.0%), high FLIPI (54.2% vs 85.0%), and high CRP (45.8% vs 70.0%). In the multivariate analysis of high-risk factors, only POD24 (hazard ratio [HR] 2.34; 95% CI, 1.02- 5.34) and high TMTV (HR 2.53; 95% CI, 1.14-5.65) were associated with shorter PFS. For pts with both POD24 and high TMTV (n=12), the CRR was 16.7% with a 12-mo PFS of 36.0%. These analyses of high-risk subgroups are exploratory in nature and should be validated in a larger study cohort.
Conclusions: With 17-mo median follow-up, tisagenlecleucel produced high ORR and CRR and was associated with durable response and promising 12-mo PFS in pts with r/r FL and 2+ prior lines of therapy. Safety was consistent with known tisagenlecleucel profile. POD24 and high TMTV were independently associated with PFS. These results suggest that tisagenlecleucel can induce high rates of durable response, including most pts in the high-risk disease subgroups, who have poor prognosis with current non-CAR-T cell therapies.
Thieblemont: Bristol Myers Squibb/Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Gilead Sciences: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses , Research Funding; Kyte: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Takeda: Honoraria, Membership on an entity's Board of Directors or advisory committees; Incyte: Honoraria, Membership on an entity's Board of Directors or advisory committees; Abbvie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Cellectis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses ; Hospira: Research Funding; Bayer: Honoraria; Amgen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Travel, Accommodations, Expenses . Dickinson: Amgen: Honoraria; Gilead Sciences: Consultancy, Honoraria, Speakers Bureau; MSD: Consultancy, Honoraria, Research Funding, Speakers Bureau; Janssen: Consultancy, Honoraria; Bristol-Myers Squibb: Consultancy, Honoraria; Takeda: Research Funding; Celgene: Research Funding; Roche: Consultancy, Honoraria, Other: travel, accommodation, expenses, Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Research Funding, Speakers Bureau. Martinez-Lopez: Incyte: Consultancy, Research Funding, Speakers Bureau; Janssen: Consultancy, Speakers Bureau; BMS: Consultancy, Research Funding, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Roche: Consultancy, Research Funding, Speakers Bureau; Astellas: Research Funding, Speakers Bureau. Kolstad: Nordic Nanovector: Membership on an entity's Board of Directors or advisory committees, Research Funding. Popplewell: Pfizer: Other: Travel; Novartis: Other: Travel; Hoffman La Roche: Other: Food. Chavez: AstraZeneca: Research Funding; Novartis: Consultancy; MorphoSys: Speakers Bureau; Karyopharm Therapeutics: Consultancy; Adaptive: Research Funding; Kite/Gilead: Consultancy; Abbvie: Consultancy; Merck: Research Funding; BeiGene: Speakers Bureau; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Bristol Myers Squibb: Speakers Bureau; Epizyme: Speakers Bureau. Bachy: Roche: Consultancy; Takeda: Consultancy; Kite, a Gilead Company: Honoraria; Novartis: Honoraria; Daiishi: Research Funding; Incyte: Consultancy. Kato: Kyowa Kirin: Honoraria, Research Funding; Janssen: Consultancy, Honoraria, Research Funding; Eisai: Consultancy, Research Funding; Dainippon-Sumitomo: Honoraria; Daiichi Sankyo: Consultancy, Research Funding; Chugai: Consultancy, Honoraria, Research Funding; Celgene: Consultancy, Honoraria, Research Funding; Bristol-Myers Squibb: Honoraria, Research Funding; AstraZeneca: Consultancy; Abbvie: Consultancy, Research Funding; MSD: Honoraria; Mundi: Honoraria; Novartis: Consultancy, Research Funding; Ono: Honoraria, Research Funding. Harigae: Novartis Pharma: Honoraria, Research Funding; Chugai Pharma: Honoraria; Janssen Pharma: Honoraria; Ono pharma: Honoraria, Other: Subsidies or Donations; Astellas Pharma: Other: Subsidies or Donations; Kyowakirin: Other: Subsidies or Donations; Bristol Myers Squibb: Honoraria. Kersten: Kite/Gilead: Honoraria, Research Funding; Novartis: Honoraria; Miltenyi Biotech: Honoraria; BMS/Celgene: Honoraria, Research Funding; Roche: Honoraria; Takeda: Honoraria. Andreadis: GenMAB: Research Funding; Karyopharm: Honoraria; Incyte: Honoraria; BMS/Celgene: Research Funding; Epizyme: Honoraria; Crispr Therapeutics: Research Funding; Atara: Consultancy, Honoraria; Novartis: Research Funding; Kite: Honoraria; Merck: Research Funding; Roche: Current equity holder in publicly-traded company, Ended employment in the past 24 months; TG Therapeutics: Honoraria. Riedell: Kite/Gilead: Research Funding, Speakers Bureau; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding; MorphoSys: Research Funding; BeiGene: Consultancy; Takeda: Consultancy, Membership on an entity's Board of Directors or advisory committees; Celgene/BMS: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Janssen: Membership on an entity's Board of Directors or advisory committees; Tessa Therapeutics: Research Funding; Bayer: Membership on an entity's Board of Directors or advisory committees; Xencor: Research Funding; Karyopharm: Consultancy, Membership on an entity's Board of Directors or advisory committees; Calibr: Research Funding. Pérez-Simón: Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding, Speakers Bureau. Chen: Mesolbast: Honoraria; Morphosys: Honoraria. Nastoupil: MorphoSys: Honoraria; Bayer: Honoraria; Genentech: Honoraria, Research Funding; Takeda: Honoraria, Other: DSMC, Research Funding; IGM Biosciences: Research Funding; Denovo Pharma: Other: DSMC; Bristol Myers Squibb/Celgene: Honoraria, Research Funding; Caribou Biosciences: Research Funding; Novartis: Honoraria, Research Funding; Gilead/Kite: Honoraria, Research Funding; TG Therapeutics: Honoraria, Research Funding; Janssen: Honoraria, Research Funding; ADC Therapeutics: Honoraria; Epizyme: Honoraria, Research Funding; Pfizer: Honoraria, Research Funding. Von Tresckow: Pentixafarm: Consultancy, Honoraria; Novartis: Consultancy, Honoraria, Other: Congress and travel support, Research Funding; MSD: Consultancy, Honoraria, Other: Congress and travel support, Research Funding; Kite-Gilead: Consultancy, Honoraria; BMS-Celgene: Consultancy, Honoraria, Other: Congress and travel support; AstraZeneca: Honoraria, Other: Congress and travel support; Amgen: Consultancy, Honoraria; AbbVie: Other: Congress and travel support; Pfizer: Consultancy, Honoraria; Roche: Consultancy, Honoraria; Takeda: Consultancy, Honoraria, Other, Research Funding. Ferreri: Gilead, Novartis, Juno, PletixaPharm, Roche, Incyte: Membership on an entity's Board of Directors or advisory committees; BMS, Beigene, Pharmacyclics, Hutchison Medipharma, Amgen, Genmab, ADC Therapeutics, Gilead, Novartis, Pfizer: Research Funding. Teshima: Fuji pharma CO.,Ltd: Research Funding; Astellas Pharma Inc.: Research Funding; TEIJIN PHARMA Limited: Research Funding; CHUGAI PHARMACEUTICAL CO., LTD.: Research Funding; Pfizer Inc.: Honoraria; Merck Sharp & Dohme: Membership on an entity's Board of Directors or advisory committees; Kyowa Kirin Co.,Ltd.: Honoraria, Research Funding; Takeda Pharmaceutical Company: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis International AG: Membership on an entity's Board of Directors or advisory committees, Other, Research Funding; Janssen Pharmaceutical K.K.: Other; Nippon Shinyaku Co., Ltd.: Research Funding; Bristol Myers Squibb: Honoraria; Sanofi S.A.: Research Funding; Gentium/Jazz Pharmaceuticals: Consultancy. Patten: ASTRA ZENECA: Honoraria; ABBVIE: Honoraria; NOVARTIS: Honoraria; GILEAD SCIENCES: Honoraria, Research Funding; ROCHE: Research Funding; JANSSEN: Honoraria. McGuirk: Kite/ Gilead: Consultancy, Honoraria, Other: travel accommodations, expense, Kite a Gilead company, Research Funding, Speakers Bureau; Novartis: Research Funding; Gamida Cell: Research Funding; Fresenius Biotech: Research Funding; Novartis: Research Funding; EcoR1 Capital: Consultancy; Magenta Therapeutics: Consultancy, Honoraria, Research Funding; Allovir: Consultancy, Honoraria, Research Funding; Juno Therapeutics: Consultancy, Honoraria, Research Funding; Astelllas Pharma: Research Funding; Bellicum Pharmaceuticals: Research Funding; Pluristem Therapeutics: Research Funding. Petzer: AppVie: Honoraria; Astra Zeneca: Honoraria; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Celgene: Honoraria; Amgen: Honoraria; Janssen: Honoraria; Sanofi: Honoraria; Takeda: Honoraria; Sandoz: Honoraria. Viardot: University Hospital of Ulm: Current Employment; F. Hoffmann-La Roche Ltd: Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis: Honoraria, Membership on an entity's Board of Directors or advisory committees; Amgen: Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Honoraria, Membership on an entity's Board of Directors or advisory committees; Kite/Gilead: Honoraria, Membership on an entity's Board of Directors or advisory committees. Zinzani: JANSSEN-CILAG: Other: Advisory board, Speakers Bureau; NOVARTIS: Consultancy, Other, Speakers Bureau; EUSAPHARMA: Consultancy, Other, Speakers Bureau; SANDOZ: Other: Advisory board; MSD: Consultancy, Other: Advisory board, Speakers Bureau; TAKEDA: Other: Advisory board, Speakers Bureau; BMS: Other: Advisory board, Speakers Bureau; TG Therapeutics: Other: Advisory board, Speakers Bureau; ROCHE: Other, Speakers Bureau; SERVIER: Other: Advisory board, Speakers Bureau; KYOWA KIRIN: Other, Speakers Bureau; Incyte: Other, Speakers Bureau; CELLTRION: Other: Advisory board, Speakers Bureau; ADC Therap.: Other; GILEAD: Other: Advisory board, Speakers Bureau; Beigene: Other, Speakers Bureau; VERASTEM: Consultancy, Other: Advisory board, Speakers Bureau. Malladi: Gilead Science: Consultancy; Gilead: Honoraria, Other: Travel support. Lobetti Bodoni: Spouse: Amgen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Spouse: Janssen: Honoraria, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Spouse: Celgene: Honoraria; Spouse: Harlcok Healthcare: Current holder of individual stocks in a privately-held company; Spouse: Takeda: Consultancy, Honoraria, Speakers Bureau; Spouse: Sanofi: Consultancy, Membership on an entity's Board of Directors or advisory committees; Spouse: NHS: Ended employment in the past 24 months; Spouse: F. Hoffmann-La Roche: Current Employment, Current equity holder in publicly-traded company; Gilead: Other: Travel sponsorship in June 2019; Novartis: Current Employment, Current equity holder in publicly-traded company. Masood: Novartis: Current Employment, Current holder of stock options in a privately-held company. Schuster: Genentech/Roche: Consultancy, Research Funding; Tessa Theraputics: Consultancy; Loxo Oncology: Consultancy; BeiGene: Consultancy; Juno Theraputics: Consultancy, Research Funding; Alimera Sciences: Consultancy; Merck: Research Funding; Incyte: Research Funding; Acerta Pharma/AstraZeneca: Consultancy; Abbvie: Consultancy, Research Funding; Novartis: Consultancy, Honoraria, Patents & Royalties, Research Funding; TG Theraputics: Research Funding; Pharmacyclics: Research Funding; Adaptive Biotechnologies: Research Funding; Nordic Nanovector: Consultancy; Celgene: Consultancy, Honoraria, Research Funding. Fowler: Bristol Myers Squibb, F. Hoffmann-La Roche Ltd, TG Therapeutics and Novartis: Membership on an entity's Board of Directors or advisory committees, Research Funding; BostonGene, Corp: Current Employment, Current holder of stock options in a privately-held company. Dreyling: Bayer HealthCare Pharmaceuticals: Consultancy, Research Funding, Speakers Bureau; Astra Zeneca: Consultancy, Speakers Bureau; BeiGene: Consultancy; Celgene: Consultancy, Research Funding, Speakers Bureau; Genmab: Consultancy; Amgen: Speakers Bureau; Incyte: Consultancy, Speakers Bureau; Gilead/Kite: Consultancy, Research Funding, Speakers Bureau; Novartis: Consultancy, Speakers Bureau; Janssen: Consultancy, Research Funding, Speakers Bureau; Roche: Consultancy, Research Funding, Speakers Bureau; Abbvie: Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal